|
April 12, 2021: Andres, Jen, Tony, and Xiao's first report on N-HVI mediated Heterocyclic Group Transfer or "HGT" entitled "Heterocyclic group transfer reactions with I(III) NHVI reagents: access to N-alkyl(heteroaryl)onium salts via olefin aminolactonization" has been accepted for publication in Chem. Sci.: https://doi.org/10.1039/D1SC00187F!
This work represents our first report on this exciting new direction for the W lab! While it may have been on Chem Rxiv for a little while now, this final report features an expanded substrate scope and a more complete picture of our platform for the synthesis of complex (heteroaryl)onium salts with N-HVIs. These salts are a versatile and emerging functional handle for organic chemistry, spanning heterocycle synthesis to radical precursors for cross coupling. With all the exciting new chemistry emerging around pyridinium salts, there has been little innovation around how we make them, and that is where we come in. Using N-HVIs, we have a highly modular and general toolkit for introducing these functional handles. In this report, we look at an "aminolactonization" from simple alkenoic acids, using in situ generated N-HVIs and operationally simple conditions. With this under our belt, we have a lot of exciting new projects underway in the group looking at sticking pyridinium salts anywhere your heart desires on organic molecules. Be sure to check it out and stay tuned!!!
1 Comment
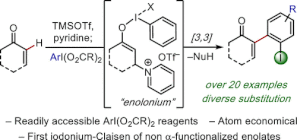
January 12, 2021: Xiao's Synpacts article examining the regioselective synthesis of Ortho-Quinones and Ortho-Quinols from Phenols using I(V) Reagents is up on Synlett!
This Perspective was an invited submission following up on our ACIE paper in 2019 on dearomatization using our I(V) Bi(N)-HVI reagents (Check that out here!). It highlights the unique utility of I(V) reagents for the regioselective ortho-oxidation of phenols, covering everything from reagent and methods development to total synthesis applications! This work outlines a novel approach to the direct C–H alpha-arylation of enones, a remaining challenge in organic synthesis. Alpha-aryl enones are valuable structural motifs and synthetic intermediates, however, perhaps surprisingly in the age of C–H functionalization, there remains no general strategy for the direct enone C–H arylation. By leveraging the unique reactivity of in situ generated b-pyridinium silyl enol ethers, Felipe and Wendy developed an arylation strategy using readily accessible ArIX2 reagents which proceeds via a reductive [3,3]-iodonium Claisen. A particularly powerful feature of this chemistry is that the entire aryl iodide moiety is transferred intact, making it highly atom economical and installing a valuable functional handle for downstream manipulations. This work builds on recent beautiful work from Shafir by extending arylative iodonium Claisen reaction to non-malonate derivatives and appears to be enabled by dual steric and electronic modulation by the beta-pyridinium moiety. This unique reactivity is the subject of ongoing investigations in our group. Congrats to Felipe (Y4) and Wendy (Temple Undergrad!) on some great work!
Xiao and Nate's paper entitled "Dearomatization of Electron-Deficient Phenols to ortho-Quinones Enabled by Bidentate Nitrogen-Ligated I(V) Reagents" has been accepted for publication in Angewandte Chemie: DOI: 10.1002/anie.201909868. This work provided the first general synthetic method for the oxidation of electron-deficient phenols, substrates that have remained elusive as they are inert to common oxidants. The resulting ortho-quinones represent valuable synthetic building blocks for complex molecule synthesis and we showcase several methods for their one-pot derivatization. Building from our lab's work with I(III) N-HVIs, this work relies on a class of bidentate nitrogen-ligated I(V) reagents, or Bi(N)-HVIs, and represents their first synthetic application. Beyond the enhanced reactivity of these Bi(N)HVIs, the presence of a bidentate nitrogen ligand results in a much more tunable platform for the design and development of I(V) reagents which our laboratory is continuing to pursue! Stay tuned!!
July 17, 2019: Myriam and Sophie's paper entitled "Chemoselective Oxidation of Equatorial Alcohols with N-Ligated Lambda-3 Iodanes" has been accepted for publication in Organic Letters: https://doi.org/10.1021/acs.orglett.9b02018)!
This work fills a remaining gap in the selective functionalization of complex scaffolds: the chemoselective oxidation of equatorial over axial alcohols! Using two mild, operationally simple procedures, the simple pyridine ligated N-HVI "Py-HVI" enables the general oxidation of alcohols to ketones with an I(III)-reagent, which offers safety and practical advantages over typical I(V) and Cr(VI) oxidants. The conformational selectivity is explored in detail and a model for predicting the relative rates of oxidation is provided. We hope that this protocol provides a new tool for complex molecule total synthesis! Congrats to Myriam and Sophie on some great work! Myriam, Felipe, Jen and Bilal all presented their work at the POCC Poster Session at UPenn Chemistry. Myriam and Jen even brought home Best Poster Prizes! Way to go ladies!
Its Official!! The W lab OG, Jen Walters, is now Jen Walters, Ph.D.! Jen has since left us for a postdoc at the Nagib Lab at The Ohio State University starting in June 2019. As the first graduate of the W lab we are so excited for her to tackle this new adventure but are a bit lost without her presence in the group! Good luck Jen, we will miss you!!!
Felipe and Brandon's paper entitled "Reactivity of (NNN)-pincer nickel(II) aryl complex towards oxidative carbon-heteroatom bond formation" has been accepted for publication in Tetrahedron in a Special Issue honoring Prof. Seth Herzon's Young Investigator Award (Accepted Article: https://doi.org/10.1016/j.tet.2018.04.029). Great work from Felipe and Brandon, and congratulations to Seth on a well deserved honor!
Jen, Tony and Aimee's paper on the ring expansion of aliphatic secondary alcohols with N-HVIs has been accepted to Eur. J. Org. Chem. DOI: 10.1002/ejoc.201800118. Not only does this method provide rapid access to challenging medium-ring oxygen heterocycles, but is applicable to the late-stage derivatization of complex natural product scaffolds, providing new opportunities in Complexity-to-Diversity pursuits. Be sure to check out the product of their great work! Congrats team!
|
Follow us on Twitter!
@TempleWLab |